It got named spin back when people were working with the Bohr model and trying to extend it to atoms beyond hydrogen. Spin is just a property that electrons (and other particles) have. For another layer, you can take the fact that you can never cool anything down to exactly 0K (-273.15C) (although you can get close) and so nothing will ever have 0 velocity. (1 value in a range of reals is like trying to throw a dart at a dartboard with an infinitely thin wire and hitting the wire). With this uncertainty, the velocity is almost definitely not 0.  
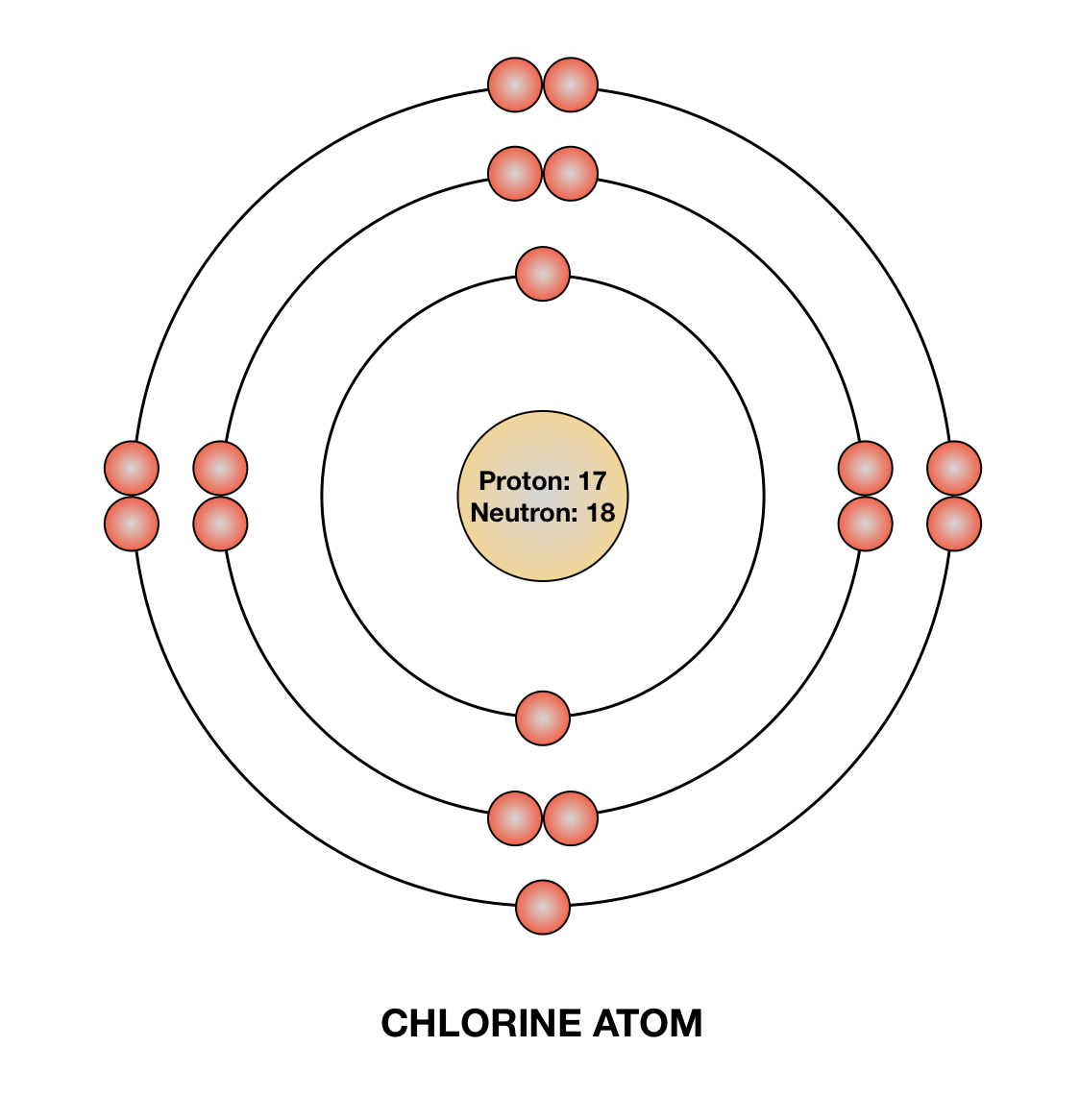
As we can't physically measure to perfect accuracy, there is an uncertainty in both measurements of the degree that we know it's probably stationary and it's probably 'over there'. So, if you know with 0 uncertainty what the velocity is, then you have no idea where it is, and all future involvement of the particle is pretty much irrelevant (how is the electron going to diffract around an atom if the electron is in a different galaxy?). The square (probability function) shows that it has an equal chance of being anywhere. If you take the infinite wavelength interpretation, then it would be nearly 0 (1/inf) but constant everywhere. Thus, the columns of the periodic table represent the potential shared state of these elements' outer electron shells that is responsible for their similar chemical characteristics.Isn't to do with the fact that the velocity is not quite 0? if you know it is exactly 0 then the uncertainty in the position is infinite as well (momentum is a function of velocity, so delta P = 0 -> delta V = 0 -> delta X = inf) therefore it has an equal probability of being anywhere. When an atom gains an electron to become a negatively-charged ion this is indicated by a minus sign after the element symbol for example, \(F^-\). Group 17 elements, including fluorine and chlorine, have seven electrons in their outermost shells they tend to fill this shell by gaining an electron from other atoms, making them negatively-charged ions. When an atom loses an electron to become a positively-charged ion, this is indicated by a plus sign after the element symbol for example, Na +. As a result of losing a negatively-charged electron, they become positively-charged ions. This means that they can achieve a stable configuration and a filled outer shell by donating or losing an electron. In comparison, the group 1 elements, including hydrogen (H), lithium (Li), and sodium (Na), all have one electron in their outermost shells. Their non-reactivity has resulted in their being named the inert gases (or noble gases). 
As shown in, the group 18 atoms helium (He), neon (Ne), and argon (Ar) all have filled outer electron shells, making it unnecessary for them to gain or lose electrons to attain stability they are highly stable as single atoms. The periodic table is arranged in columns and rows based on the number of electrons and where these electrons are located, providing a tool to understand how electrons are distributed in the outer shell of an atom. Elements in other groups have partially-filled valence shells and gain or lose electrons to achieve a stable electron configuration.Īn atom may gain or lose electrons to achieve a full valence shell, the most stable electron configuration. A full valence shell is the most stable electron configuration. Group 18 elements (helium, neon, and argon are shown) have a full outer, or valence, shell. \):īohr diagrams indicate how many electrons fill each principal shell.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
